

SOP-Guard
The efficiency booster
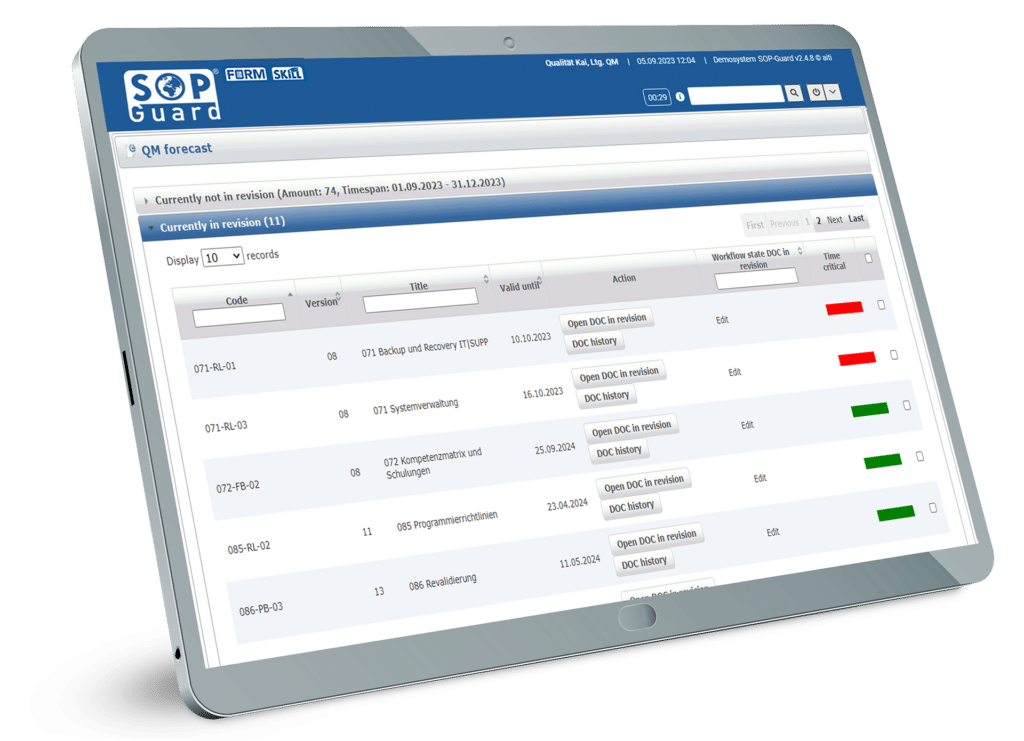
SOP-Guard document management
Realise the full potential of your administrative tasks in quality management and optimise your workflows with the innovative support of SOP-Guard. Regardless of your industry, SOP-Guard makes creating, reviewing, approving, distributing, and archiving documents easy. The integrated document training gives you user-based control over your electronic document management for GxP systems. Using SOP-Guard paves the way for a future of efficiency, productivity and success.



Top 5 benefits
- Validated software for 100% electronic document management in regulated companies
- Precise monitoring of deadlines, including timely corrective measures.
- Correct mapping of competencies and responsibilities.
- Automated assignment of documents to the right user group.
- Automation of previously manual work steps in document management.
SOP-Guard is validated according to GAMP 5, GxP and FDA compliant.
Customer Testimonials
The low effort involved in creating, checking, approving, managing and training a wide variety of specification documents and the flexibility of SOP-Guard were clear plus points.
Dr. Martina MaritzHead Corporate Quality Assurance Develco Pharma GmbH
A DMS can cover three quarters of our requirements, but the spice lies in the remaining 25 percent that SOP-Guard offers us.
Ivica SaricLeiter IT | Validierungskoordinator ITUrsapharm Arzneimittel GmbH
SOP-Guard is easy to use and quick to understand. This has considerably simplified the roll-out in six Eastern European countries.
Mag.pharm. Georg E. SchmidtHead of Pharmaceutical AffairsChiesi Pharmaceuticals GmbH
SOP Guard makes life much easier for us in a smaller academic GMP facility. Simple operation, logical structure, fast customer service and well-prepared documents for validation and revalidation of the system make it a very helpful program for efficient and GMP-compliant document control.
Dr. Daniela WehlerSachkundige PersonIII. Med. Klinik und Poliklinik, Universitätsmedizin Mainz
Quick-Facts
Explicitly designed for GxP environments, SOP-Guard provides a centralised platform for easy management and control of essential documents. The specialised and powerful programme features, combined with an intuitive user interface, reduce errors and wasted time. SOP-Guard fully complies with current regulatory requirements, providing improved efficiency, increased security and better document control.
We attach great importance to feedback from our users and customers developing our software. Our solution has received numerous positive reviews and successfully passed audits and inspections. Let us help you improve your document management processes for the success of your organisation.
Our software solution can be installed at a location of your choice (on-site, private cloud, etc.) so that both the software and the data storage take place within your company’s data sovereignty. Thanks to web technology, our software is compatible with standard devices such as PCs, laptops, and tablets. The software comes to you as a validated virtual machine, and our maintenance contract strictly defines our responsibilities and clarifies our respective roles. In addition, we offer numerous interfaces that enable seamless connectivity with existing systems (e.g. Active Directory via LDAP).
- SOP-Guard is fully validated as a product in accordance with GAMP 5 (category 3).
- The software fulfils the requirements of the following regulations, among others: GxP (EU), ICH and 21 CFR Part 11.
- Traceability through system-internal audit trail for detailed recording of actions in the system
- Comprehensive validation packages (installation documentation, test case documents, risk analysis …)
SOP-Guard explained quickly
Downloads
SOP-Guard
| Title | Categories | Update Date | Download |
|---|---|---|---|
|
SOP Guard: Dokumentenmanagement – modern & GxP konform |
Softwarelösungen, SOP-Guard | 2023/09/19 | Download |









